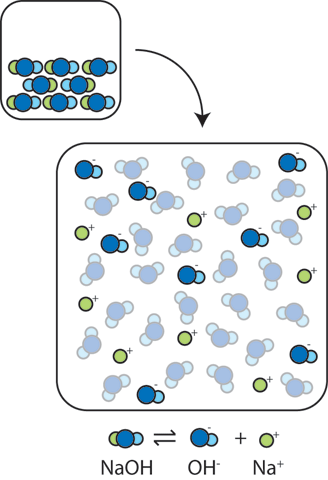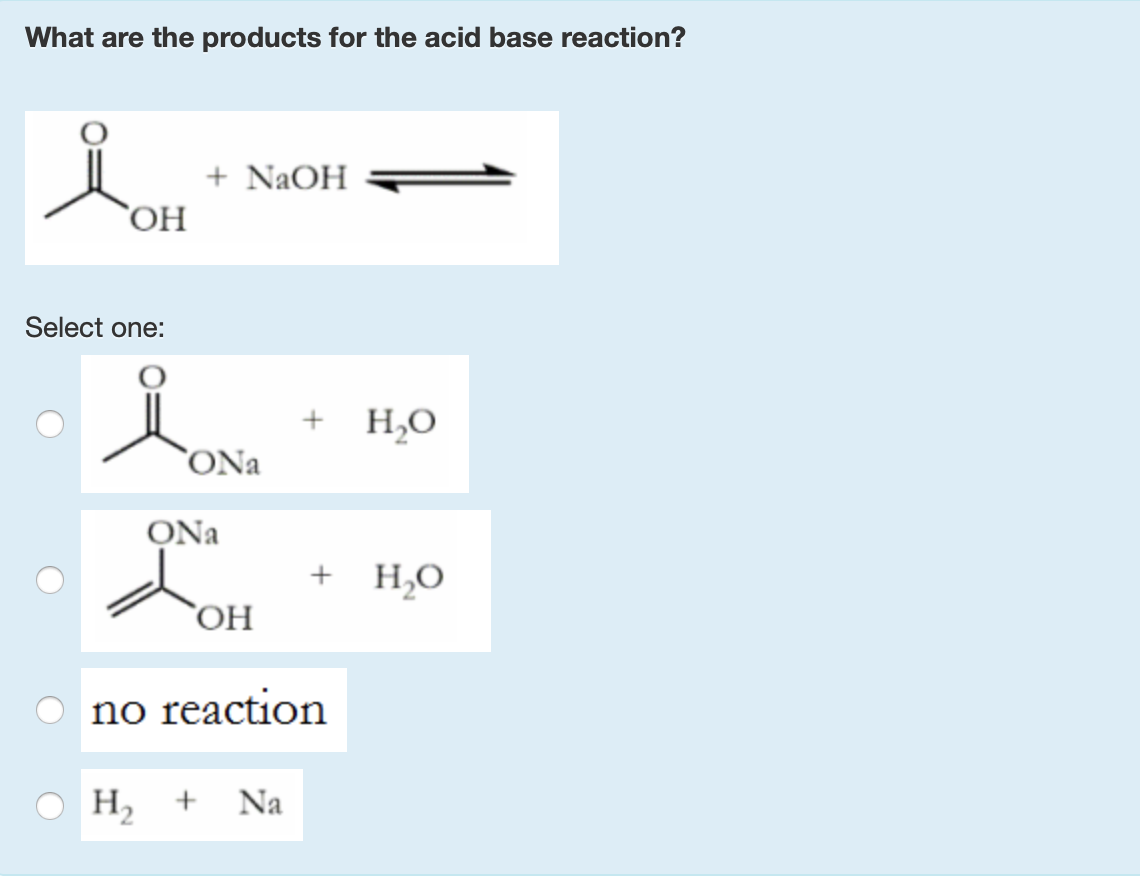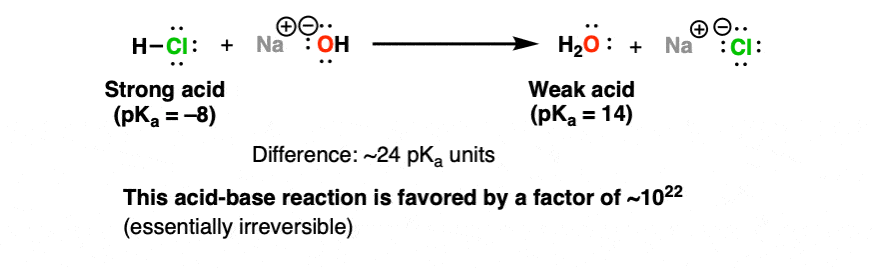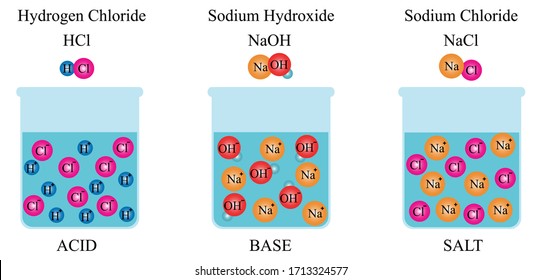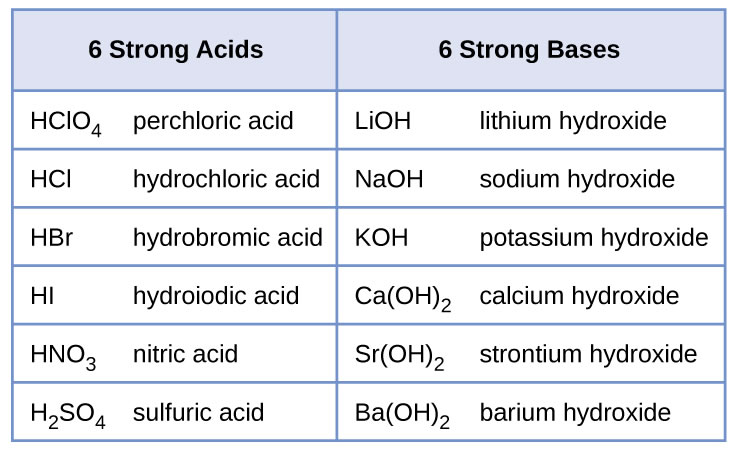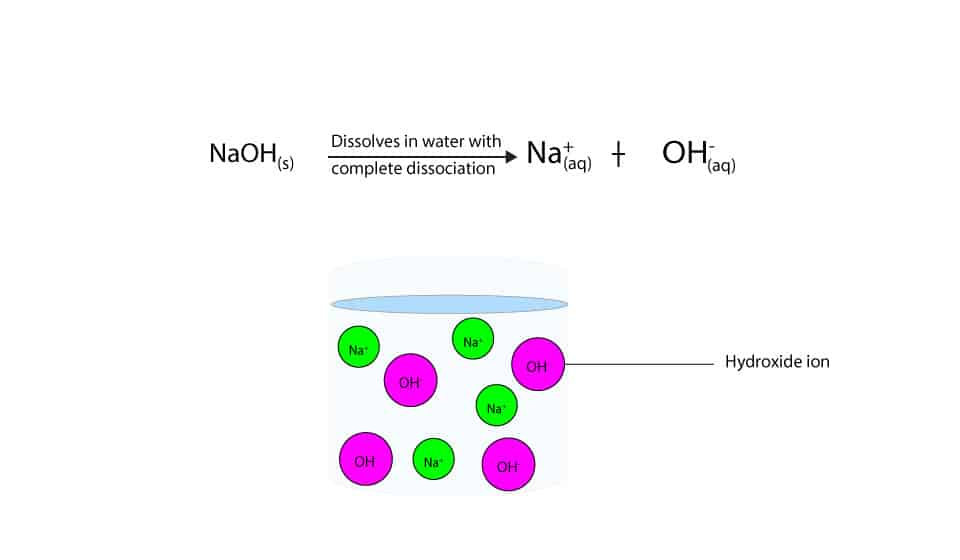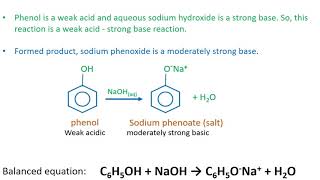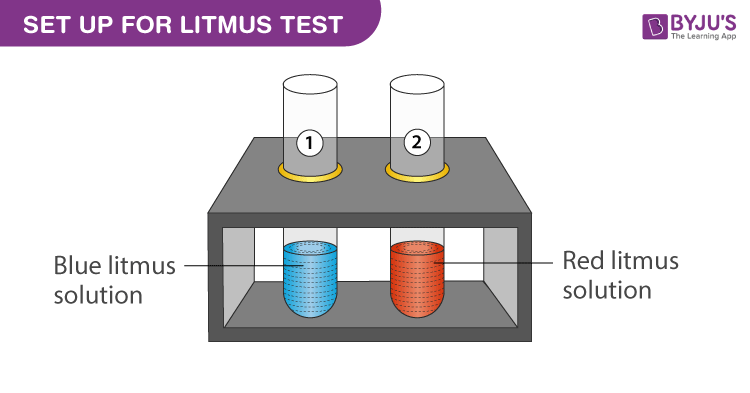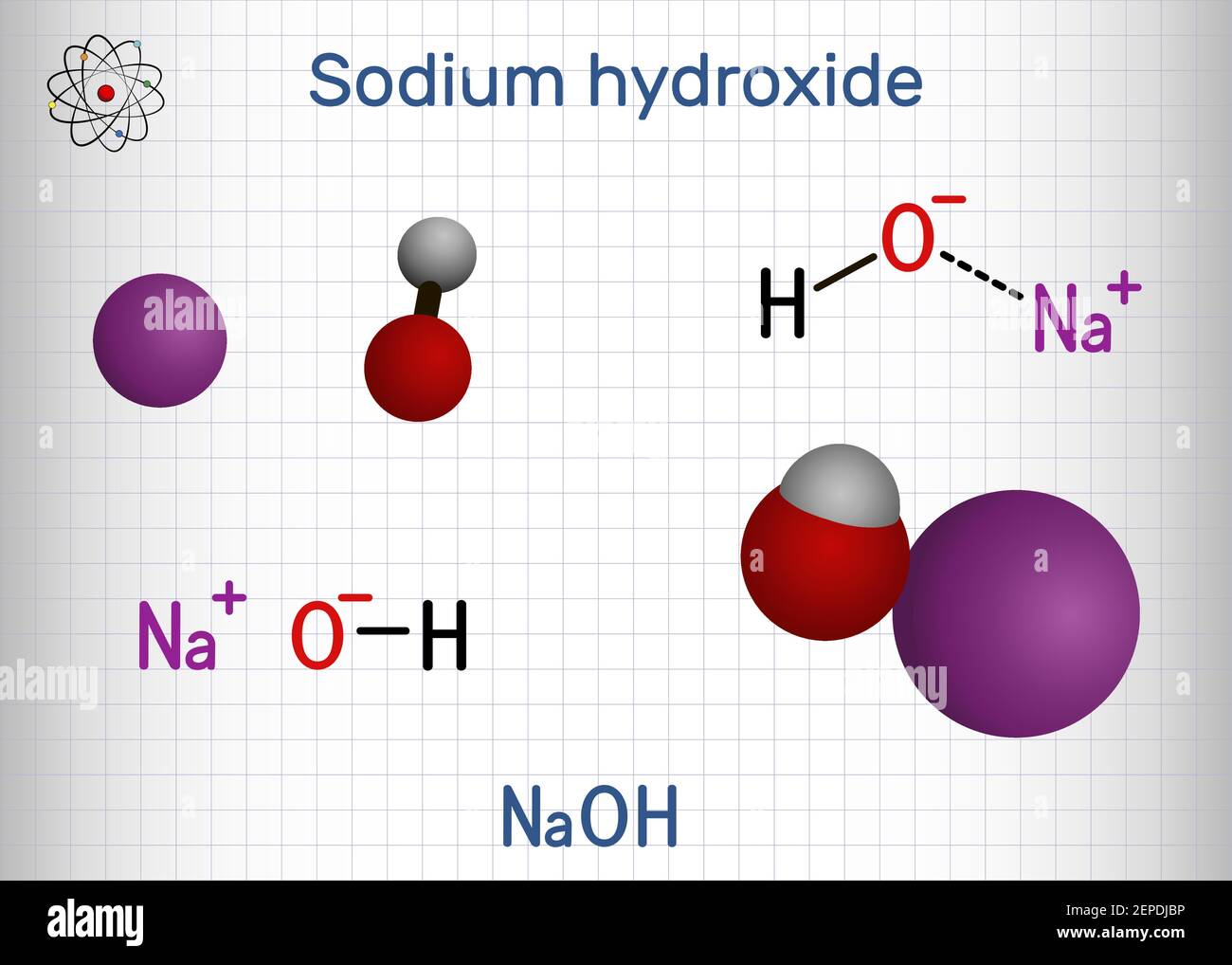
Sodium hydroxide, caustic soda, lye molecule. NaOH is highly caustic base and alkali, ionic compound. Structural chemical formula and molecule model Stock Vector Image & Art - Alamy

Selective focus of sodium hydroxide base and sulfuric acid solution in brown glass and plastic bottle inside a chemistry laboratory. White background Stock Photo - Alamy



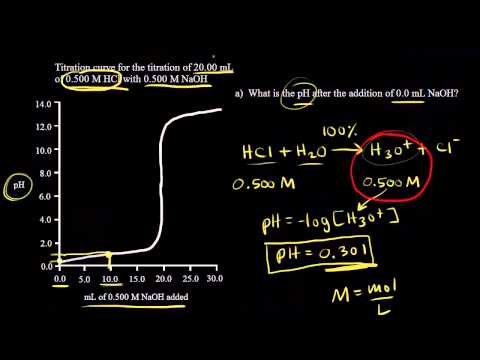




:max_bytes(150000):strip_icc()/prepare-sodium-hydroxide-or-naoh-solution-608150_FINAL-696b52d6f90b4b1383ec8f95db73a1f3.png)



-in-water-01.jpg)